NGM282, NAS and fibrosis in NASH:
a phase 2 study
Harrison SA, AASLD 2018, Abs. 104
Design
* NAS (NAFLD Activity Score): steatosis (0 to 3), lobular inflammation (0 to 3), ballooning (0 to 2)
- NGM282: engineered variant of human FGF19
- Rosuvastatin: started at W2 if LDL-cholesterol rise ≥ 10 mg/dl
Endpoints
- Primary: decrease in absolute liver fat content (MRI-PDFF) ≥ 5% at W12
- Exploratory: change in liver histology at W12
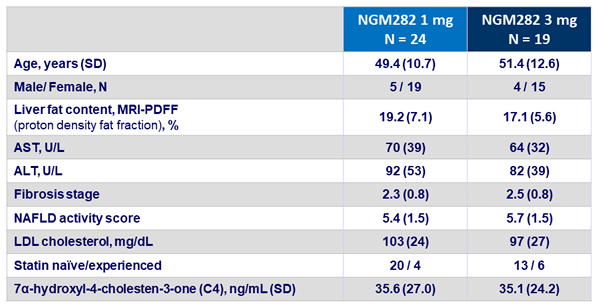
Baseline characteristics, mean (SD)
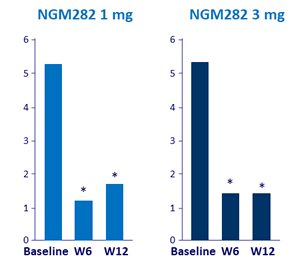
Mean C4 levels (ng/ml)
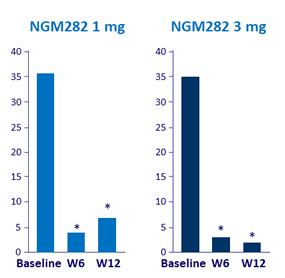
* p < 0.001 vs baseline
Mean total serum bile acids (µmol/L)
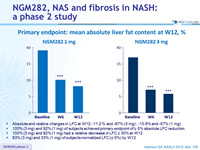
Primary endpoint: mean absolute liver fat content at W12, %
- Absolute and relative changes in LFC at W12: -11.2 % and -67% (3 mg) ; -10.9% and -57% (1 mg)
- 100% (3 mg) and 92% (1 mg) of subjects achieved primary endpoint of ≥ 5% absolute LFC reduction
- 100% (3 mg) and 92% (1 mg) had a relative decrease in LFC ≥ 30% at W12
- 63% (3 mg) and 33% (1 mg) of subjects normalized LFC (≤ 5%) by W12
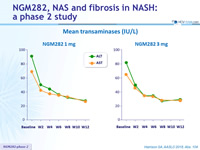
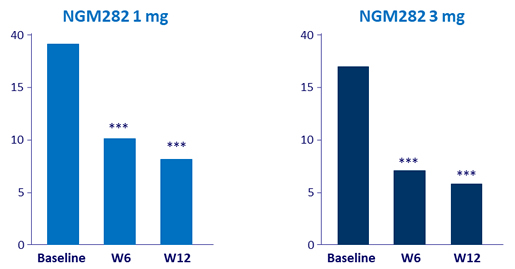
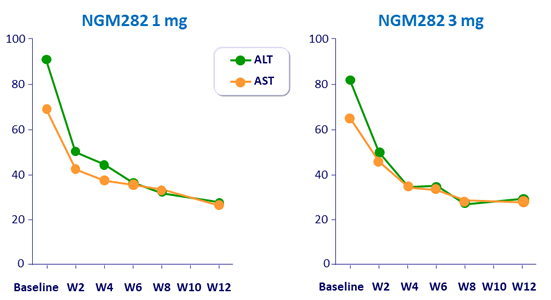
Mean transaminases (IU/L)
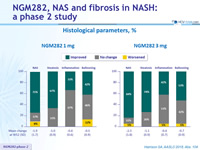
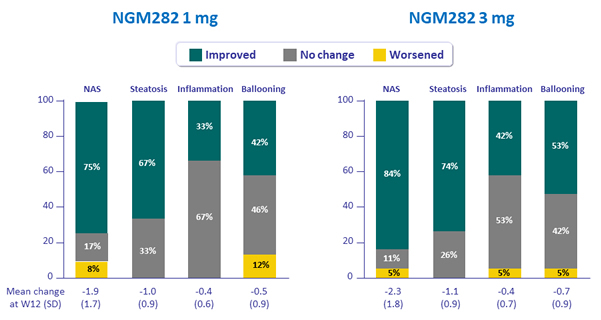
Histological parameters, %
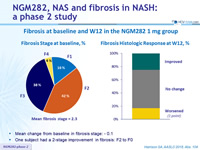
Fibrosis at baseline and W12 in the NGM282 1 mg group

- Mean change from baseline in fibrosis stage: - 0.1
- One subject had a 2-stage improvement in fibrosis: F2 to F0
Safety and tolerability
- Favorable safety and tolerability profile consistent with other NGM282 studies: no new safety signals identified
- Mild gastrointestinal symptoms (loose/frequent stools) were the most common treatment emergent adverse events
- Majority were mild and resolved during treatment phase
- No subject withdrew from treatment due to any drug-related AEs
- Gastrointestinal symptoms were largely mitigated with separating the timing of injection around meals and decreasing meal size
- Five severe adverse events, all unrelated to study drug:
- Pneumonia
- Pleurisy
- Chest tightness
- Cardiac arrest (non myocardial infarction)
- Renal mass
Summary
- Potent C4 and bile acid suppression consistent with FGF19 hormone activity
- Significant and clinically meaningful reductions across non-invasive markers of NASH-related disease
- Large percentage of patients demonstrated histological improvement at W12
- Statin co-administration rapidly mitigates LDL-cholesterol elevations
- Treatment was safe and well tolerated