SURVEYOR-I Study - Part 2: glecaprevir + pibrentasvir in genotype 1, 4, 5, 6 – Phase II
Kwo PY. J Hepatol 2017; 67 :263-71
Anti-HCV
Glecaprevir (ABT-493)
Pibrentasvir (ABT-530)
Glecaprevir (ABT-493)
Pibrentasvir (ABT-530)
Genotype
2
3
2
3
Treatment history
Naive
IFN-Experienced
Naive
IFN-Experienced
Cirrhosis
Yes
No
Yes
No
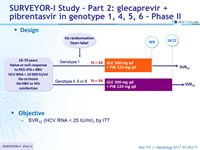
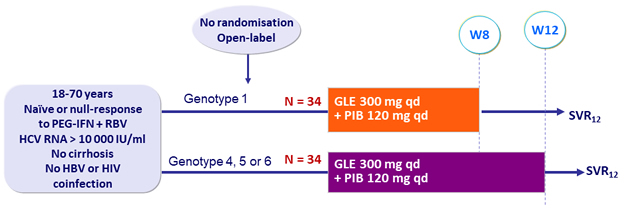
Design
Objective
- SVR12 (HCV RNA < 25 IU /ml), by ITT
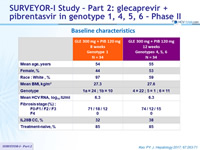
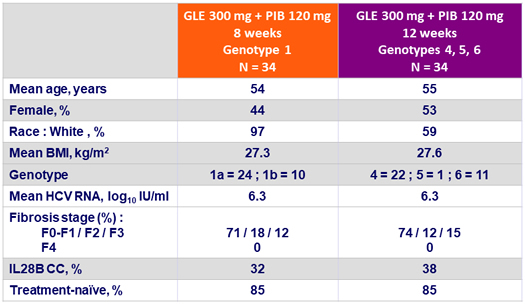
Baseline characteristics
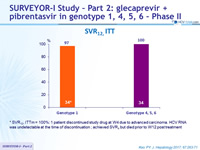
SRV12, ITT
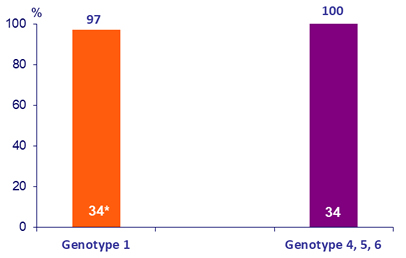
* SVR12, ITTm = 100%: 1 patient discontinued study drug at W4 due to advanced carcinoma. HCV RNA was undetectable at the time of discontinuation ; achieved SVR 4 but died prior to W12 post treatment
Resistance analysis (population sequencing with 15% threshold)
- Genotype 1 : baseline RASs in 76%: NS3 only in 48%,NS5A only in 15%, NS3 + NS5A in 12%
- Genotype 4, 5, 6: 5/22 genotype 4 had baseline NS5A RASs, and 7/11 genotype 6 patients had baseline RASs (NS3 only in 2,NS5A only in 4, NS3 + NS5A in 1)
- No impact of baseline RASs on SVR12
Adverse events and laboratory abnormalities, %

Summary
- High SVR rates were achieved in HCV genotype 1-infected patients with once daily combination of GLE + PIB
- By ITTm , all patients without cirrhosis achieved SVR12 after 8 weeks of treatment (97% by ITT)
- No impact on efficacy of baseline NS3 and/or NS5A RAVs
- 100 % SVR12 was achieved in patients with HCV genotype 4, 5 or 6 following 12 weeks of GLE + PIB
- Adverse events were mostly mild in severity
- No discontinuation for adverse event related to study drug