ENDURANCE-1 Study: glecaprevir/pibrentasvir in genotype 1 without cirrhoss
Zeuzem S. NEJM 2018; 378:354-69
Glecaprevir (ABT-493)
Pibrentasvir (ABT-530)
1
Naive
IFN-Experienced
No
HIV co-infection
Design
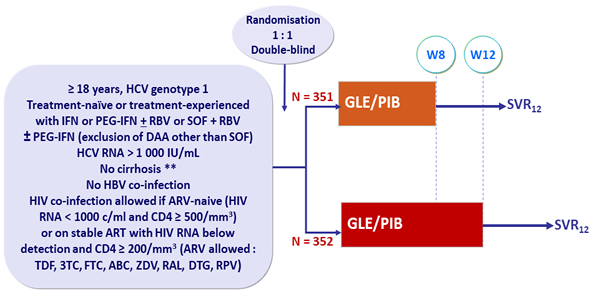
* Fibroscan® < 12.5 kPa or FibroTest® ≤ 0.48 + APRI < 1
- GLE/PIB: 100/40 mg 3 tablets QD
Objectives (SVR12)
- Non-inferiority of the 12-week regimen, by ITT-PS (exclusion of HIV and prior SOF), with lower margin of the 2-sided CI > 91% (historical rate)
- Non-inferiority of the 12-week regimen, by ITT-PS-PP (ITT-PS + exclusion of premature discontinuation or virologic failure prior to W8 and missing data in the SVR12 window), with lower margin of the 95% CI for the difference = 5%
- Non-inferiority of the 8-week regimen, by ITT-PS, with a lower margin of the 95% CI for the difference = 5%
- Secondary endpoints
- Efficacy in HIV-co-infection and in patients with prior SOF treatment, by ITT
- Virologic failure and relapse
- Resistance analysis (15% detection threshold)
- Safety
PS = Primary Subset
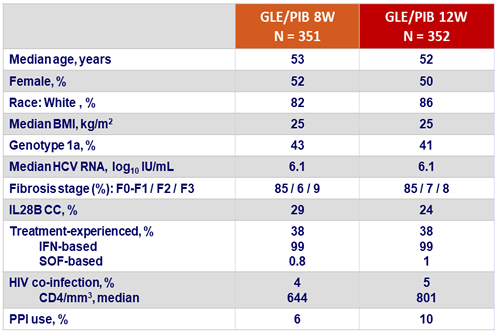
Baseline characteristics
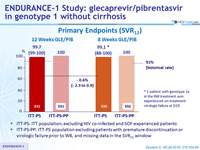
Primary Endpoints (SVR12)
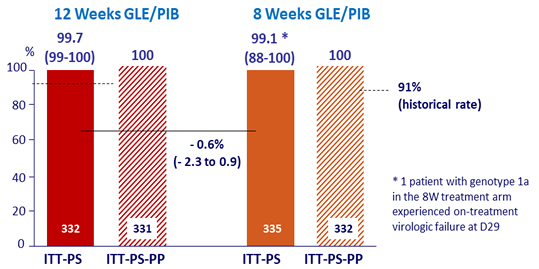
ITT-PS: ITT population, excluding HIV co-infected and SOF-experienced patients
ITT-PS-PP: ITT-PS population excluding patients with premature discontinuation or virologic failure prior to W8, and missing data in the SVR12 window
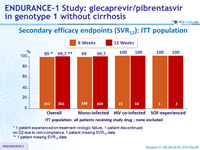
Secondary efficacy endpoints (SVR12): ITT population
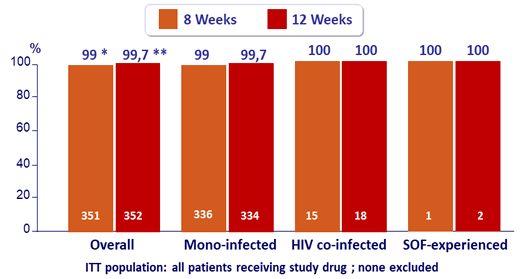
* 1 patient experienced on-treatment virologic failure, 1 patient discontinued on D2 due to non-compliance, 1 patient missing SVR12 data
** 1 patient missing SVR12 data
Adverse events and laboratory abnormalities, N (%)
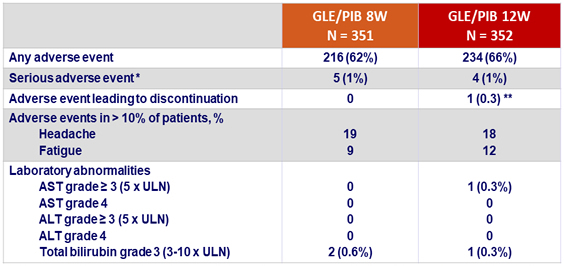
* On treatment: pneumonia aspiration, atrial fibrillation, angina unstable, radius fracture, transient ischemic attack, irritable bowel syndrome. Post-treatment: bronchitis, uterine myoma, suicide attempt.
** dandruff, anxiety and amnesia, all deemed not related to therapy.
1 death occurred during post-treatment period due to an unknown cause considered unrelated to study drug
Summary
- 99-100% of genotype 1-infected patients without cirrhosis achieved SVR12 with 8 or 12 weeks of glecaprevir / pibrentasvir (GLE/PIB)
- 8-week treatment was non-inferior to 12-week treatment (all 3 primary endpoints were met)
- SVR12 rates were high regardless of
- HIV-1 co-infection (limitation: 5% of the study population)
- prior treatment experience
- baseline HCV RNA
- presence of baseline polymorphisms
- or other factors
- GLE/PIB was well tolerated
- Only 1 patient (0.1%) discontinued study drugs for adverse event
- No significant laboratory abnormalities