SAPPHIRE-I Study: ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin for genotype 1
Treatment of HCV with ABT-450/r–Ombitasvir and Dasabuvir with Ribavirin
Feld JJ. NEJM 2014;370:1594-1603
Anti-HCV
Ombitasvir
Paritaprevir/ritonavir
Dasabuvir
Ribavirin
Ombitasvir
Paritaprevir/ritonavir
Dasabuvir
Ribavirin
Genotype
1
1a
1b
1
1a
1b
Treatment history
Naive
Naive
Cirrhosis
No
No
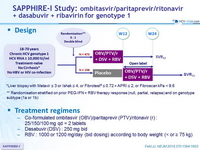
Design
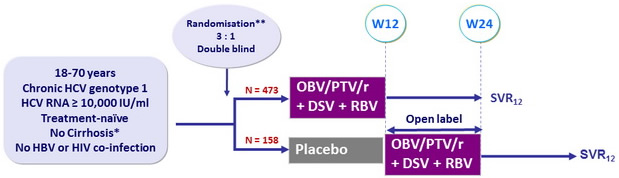
* Liver biopsy with Metavir ≤ 3 or Ishak ≤ 4, or Fibrotest® ≤ 0.72 + APRI ≤ 2, or Fibroscan kPa < 9.6
** Randomisation stratified on prior PEG-IFN + RBV therapy response (null, partial, relapse) and on genotype subtype (1a or 1b)
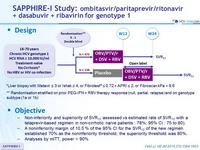
Treatment regimens
- Co-formulated ombitasvir (OBV)/paritaprevir (PTV)/ritonavir (r) : 25/150/100 mg qd = 2 tablets
- Dasabuvir (DSV) : 250 mg bid
- RBV : 1000 or 1200 mg/day (bid dosing) according to body weight (< or ≥ 75 kg)
Objectives
- Non-inferiority and superiority of SVR12 assessed vs estimated rate of SVR12 with a telaprevir-based regimen in non-cirrhotic naïve patients : 78%; 95% CI : 75 to 80).
- A noninferiority margin of 10.5 % of the 95% CI for the SVR12 of the new regimen established 70% as the noninferiority threshold; the superiority threshold was 80%.
- Analyses by mITT, power > 90%
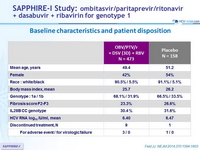
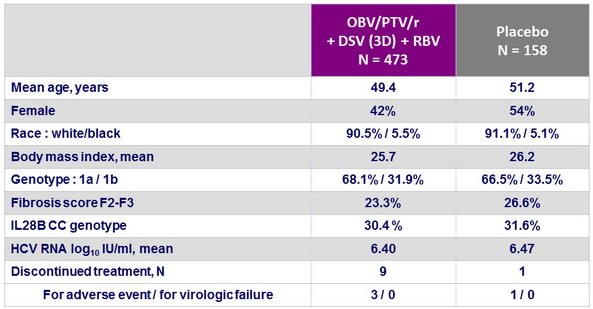
Baseline characteristics and patient disposition
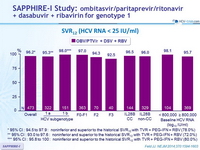
SVR12 (HCV RNA < 25 IU /ml)
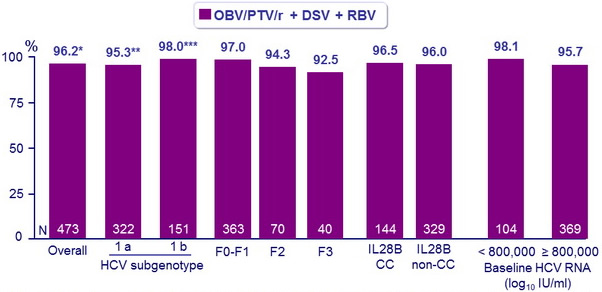
* 95% CI : 94.5 to 97.9 : noninferior and superior to the historical SVR12 with TVR + PEG-IFN + RBV (78.0%)
** 95% CI : 93.0 to 97.6 : noninferior and superior to the historical SVR12 with TVR + PEG-IFN + RBV (72.0%)
*** 95% CI : 95.8 to 100 : noninferior and superior to the historical SVR12 with TVR + PEG-IFN + RBV (80.0%)
Virologic failure
- N = 1, at W12 (genotype 1a)
Relapse
- N = 7 (1.5%)
- 5 by W4 post-treatment
- 2 by W12
Resistance testing (population sequencing) of the 7 relapses + 1 failure
- All had ≥ 1 mutant resistant variants
- Genotype 1a (N = 7) : D168V (N = 6) in NS3 ; M28T (N = 2) and Q30R (N = 3) in NS5A ; S556G (N = 3) in NS5B
- Genotype 1b (N = 1) : Y56H + D168V (NS3), L31M + Y93H (NS5A) and S556G (NS5B)
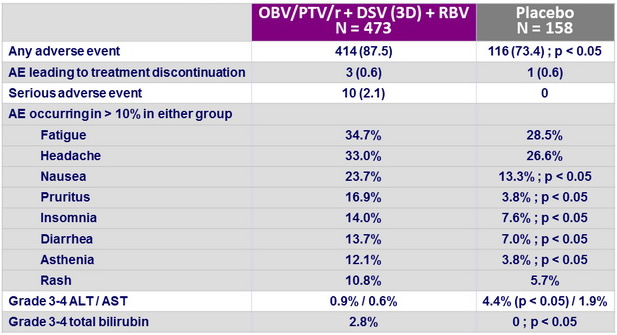
Adverse events, N (%)
Summary
- Rates of response to a 12-week interferon-free combination regimen, of ombitasvir/paritaprevir/ritonavir + dasabuvir + ribavirin, were more than 95% among previously untreated patients with HCV genotype 1 infection
- SVR12 was non inferior and superior to the historical control rate with telaprevir plus PEG-IFN + RBV in a similar patient population
- SVR12 was similar in patients with HCV genotype 1a or 1b infection, and in various subgroups (age, sex, fibrosis, IL28B)
- Tolerability was good, with
- 0.6% of patients discontinuing for adverse event
- Nausea, pruritus , insomnia, diarrhea, and asthenia more frequent in the active group
- Low incidence of grade 3-4 bilirubin elevation
- In conclusion, a 12-week all-oral combination regimen of OBV/PTV/r + DSV + RBV resulted in SVR12 > 95%, regardless of HCV genotype (1a or 1b) and with low rates of treatment discontinuation, in previously untreated patients with HCV genotype 1 infection and no cirrhosis