ASTRAL-3 study: SOF/VEL vs SOF + RBV in genotype 3
ASTRAL-3 study: SOF/VEL vs SOF + RBV in genotype 3
Foster GR. N Engl J Med 2015; 373: 2608-17
Anti-HCV
Velpatasvir (GS-5816)
Sofosbuvir
Ribavirin
Velpatasvir (GS-5816)
Sofosbuvir
Ribavirin
Genotype
3
3
Treatment history
Naive
IFN-Experienced
Naive
IFN-Experienced
Cirrhosis
Yes
No
Yes
No
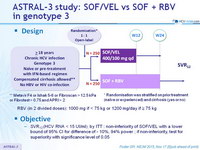
Design
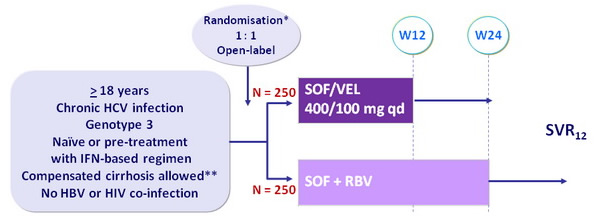
* Randomisation was stratified on prior treatment (naïve or experienced) and cirrhosis (yes or no)
** Metavir F4 or Ishak 5-6 or Fibroscan > 12.5 kPa or Fibrotest > 0.75 and APRI > 2
- RBV (in 2 divided doses) : 1000 mg if < 75 kg or 1200 mg/day if = 75 kg
Objective
- SVR12 (HCV RNA < 15 UI/ml) , by ITT : non-inferiority of SOF/VEL with a lower bound of 95% CI for difference of - 10%, 94% power ; if non-inferiority, test for superiority with significance level of 0.05
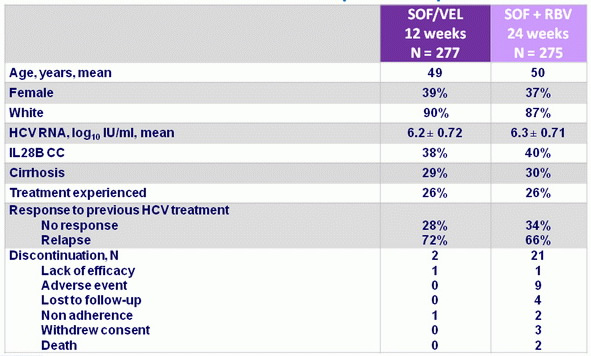
Baseline characteristics and patient disposition
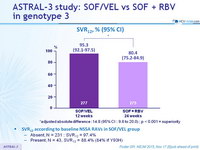
SVR12, % (95% CI)
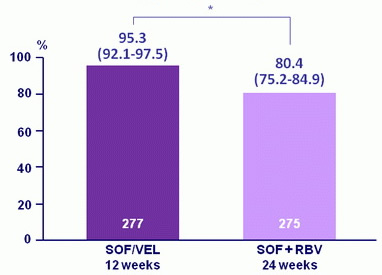
*adjusted absolute difference : 14.8 (95% CI : 9.6 to 20.0) ; p < 0.001 = superiority
SVR12 according to baseline NS5A RAVs in SOF/VEL group
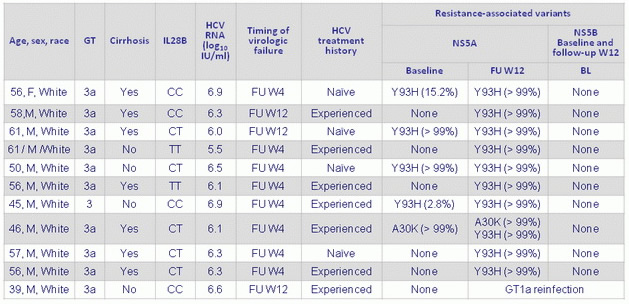
- Absent, N = 231 : SVR12 = 97.4%
- Present, N = 43, SVR12 = 88.4% (84% if Y93H)
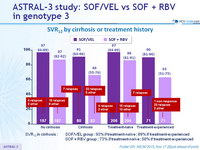
SVR12 by cirrhosis or treatment history
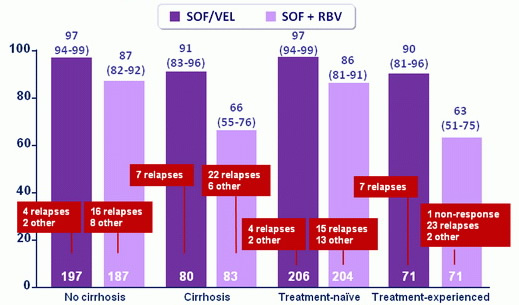
SVR12 in cirrhosis :
- SOF/VEL group : 9 3 % if treatment-naïve ; 89% if treatment-experienced
- SOF + RBV group : 73% if treatment-naïve ; 58% if treatment-experienced
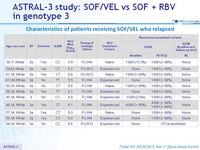
Characteristics of patients receiving SOF/VEL who relapsed
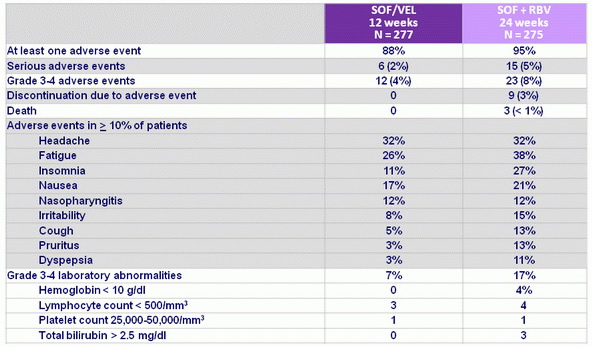
Adverse events, N (%)
Summary
- Rates of SVR12 in every subgroup of patients with HCV genotype 3 were substantially higher among those who had received 12 weeks of SOF/VEL compared to 24 weeks of SOF + RBV, including patients with cirrhosis and previous treatment failure
- Overall SVR12 of 95% with SOF/VEL for 12 weeks versus 80% with SOF + RBV for 24 weeks (p < 0.001 )
- 91% SVR12 rate in patients with cirrhosis
- Limitation : small number of black patients
- However, the rate of SVR12 was 88% among patients who had NS5A RAVs at baseline and 97% among those who did not, with the lowest rate (84%) observed among patients with the Y93H variant at baseline
- SOF/VEL was well tolerated and, compared with SOF + RBV, lacked toxicities commonly associated with RBV
- For patients with HCV genotype 3 infection, SOF/VEL for 12 weeks represents an improvement over standard treatment with 24 weeks of SOF + RBV, with a simple and highly effective regimen, together with shorter duration of treatment and fewer side effects, owing to the removal of RBV from the regimen